Regulatory consultancy specialized for national and international laboratories
We operate with technical independence, absolute confidentiality, and health responsibility, positioning ourselves as a Trusted Regulatory Partner for organizations seeking excellence, institutional security, and a long-term vision.
Our services
Registry of medicines
We manage the process for obtaining the Sanitary Registration of Generic, Herbal, Vitamin, Biological and Biotechnological Medicines, integrating the dossier and comprehensively coordinating the regulatory strategy for their authorization and commercialization
Food supplements
We manage the classification consultation for dietary supplements and provide comprehensive advice on label preparation and review, ensuring compliance with current COFEPRIS regulations, guidelines, and criteria, guaranteeing regulatory compliance for their proper marketing in Mexico




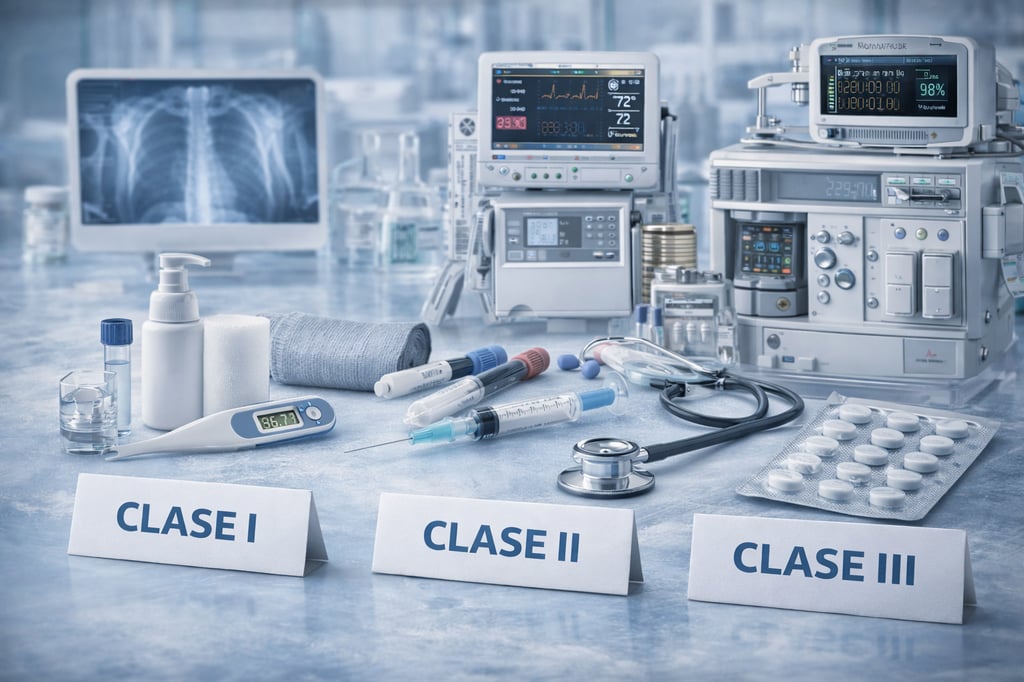
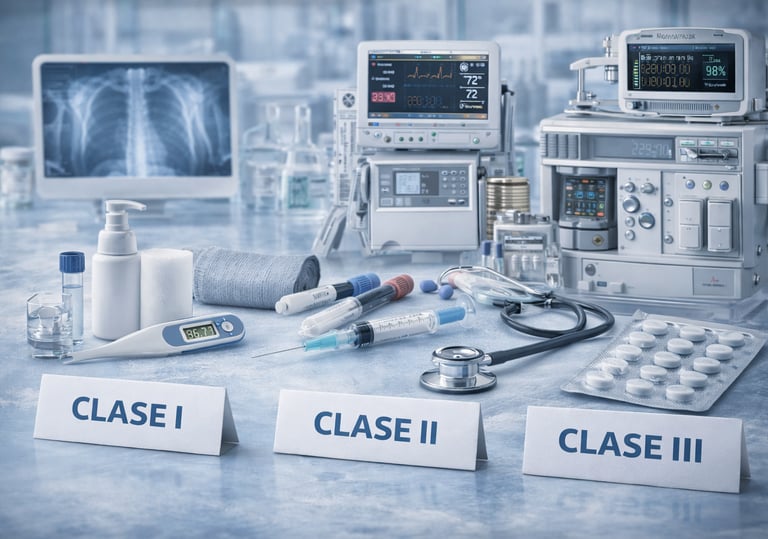
Medical devices
We manage the process for obtaining the Sanitary Registration of Class I, Class II and Class III Medical Devices, coordinating the regulatory strategy and the integration of the technical file to guarantee compliance with the applicable regulatory requirements
Import
We comprehensively manage the import procedures for regulated products, from obtaining the Import Health Permit to coordinating the customs release process, ensuring compliance with health and customs regulations




Trademark Registration
We manage trademark registration with the IMPI, incorporating feasibility analysis, name availability studies, and strategic management of the entire process until its resolution
Incorporation of companies
We structure and manage the incorporation of companies in Mexico, with a specialized focus and comprehensively coordinating the legal, corporate and regulatory aspects




Additional services
Storage and distribution warehouses
We offer a comprehensive service for warehouses and distribution centers, focused on the implementation and strengthening of the Quality Management System, including the development of the Quality Manual, Standard Operating Procedures, and all the necessary documentation for compliance with Good Distribution Practices (GDPs)
Pharmacies
We provide a comprehensive service for pharmacies, including the development of procedures, controls and documentation necessary for compliance with applicable health regulations and good storage and dispensing practices




Anser Pharma
Regulatory Consultancy and Pharmaceutical Solutions
Contact
soporte@anserpharma.com
+52 552 396 9059
© 2026. All rights reserved.


